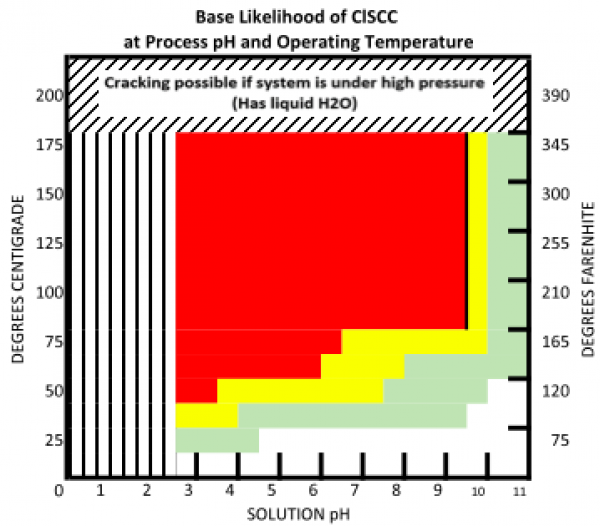
Chloride Stress Corrosion Cracking (Cl-SCC) is a form of stress corrosion cracking (SCC) caused by the combined effects of tensile stress (applied or residual), temperature, and an aqueous chloride environment. Chloride SCC is characterized by brittle-looking surface cracks with many branches. The cracking is usually transgranular, but in sensitized 300 series stainless steels it can be intergranular. Austenitic (e.g., 300 series) stainless steels are generally most prone to chloride SCC, and while duplex stainless steels and nickel base alloys are typically more resistant, they are not strictly immune (nickel content of the alloy has a notable effect on overall resistance). Carbon steels, low alloy steels, and 400 series stainless steels are not susceptible to chloride SCC.
Despite the fact that chloride SCC is one of the most well-known forms of stress corrosion cracking, it continues to plague the refining and chemical processing industries. This is typically due to inadvertent contamination of equipment with chlorides that was not anticipated by design engineers who are unaware of the potential consequences of using austenitic stainless steels where chlorides may be present. Fortunately, because of the very high toughness of stainless steel, catastrophic failures from chloride SCC are rare, although they can happen. The consequences from most leaks tend to be economic in nature, although this can still be devastating to some plants due to the high costs associated with replacing equipment.
Areas Susceptible to Chloride SCC
Similar to other SCC damage mechanisms, non-post weld heat treated (PWHT) weld regions are usually the most susceptible to chloride SCC damage due to elevated tensile residual stresses, and cold worked components, such as expansion bellows, are areas of concern due to residual plastic strains. Other common locations that tend to exhibit chloride SCC include water-cooled condensers and crude tower overhead condensers. Stainless steel drains in hydroprocessing units or boilers are also at risk, particularly during start-up if they are not purged correctly. Solid stainless steel ring-type joints (RTJs) on flanges (often used on hydroprocessing reactors or heat exchangers) and instrument tubing are also common locations of damage. Biofuel/renewable units are also prone to chloride SCC because of the often-high levels of organic chlorides in feedstocks that get converted to inorganic chlorides in reactor effluent
Chloride SCC is covered in more detail in API RP 571 - Damage Mechanisms Affecting Fixed Equipment in the Refining Industry.
References
- Reynolds, J., 2003, “99 Diseases of Pressure Equipment - Part 3,” Inspectioneering Journal, 9(6), pp. 8-11.
- Prueter, P., 2022, “Damage Control: Stress Corrosion Cracking Detection,” Inspectioneering Journal, 28(2), pp. 45-52.
Related Topics
- Amine Stress Corrosion Cracking
- Ammonia Stress Corrosion Cracking
- Carbonate Stress Corrosion Cracking
- Caustic Stress Corrosion Cracking
- Polythionic Acid Stress Corrosion Cracking (PASCC)
Relevant Links
Topic Tools
Share this Topic
Contribute to Definition
We welcome updates to this Integripedia definition from the Inspectioneering community. Click the link below to submit any recommended changes for Inspectioneering's team of editors to review.
Contribute to Definition